Location preferences
Close
Currency dollar ($)
Ava is an innovative technology company advancing women’s reproductive health.

Ava’s claims are backed by rigorous clinical trials. The results are published in peer-reviewed medical journals and contribute to the scientific body of knowledge around female reproductive health.
Our data science team draws on results from clinical trials along with the more than 3.5-million cycles recorded by our users to continually refine Ava’s machine-learning algorithms.
Advanced hardware and software interact seamlessly, translating the data recorded by Ava’s wearable sensor bracelet to its comprehensive in-app user interface.
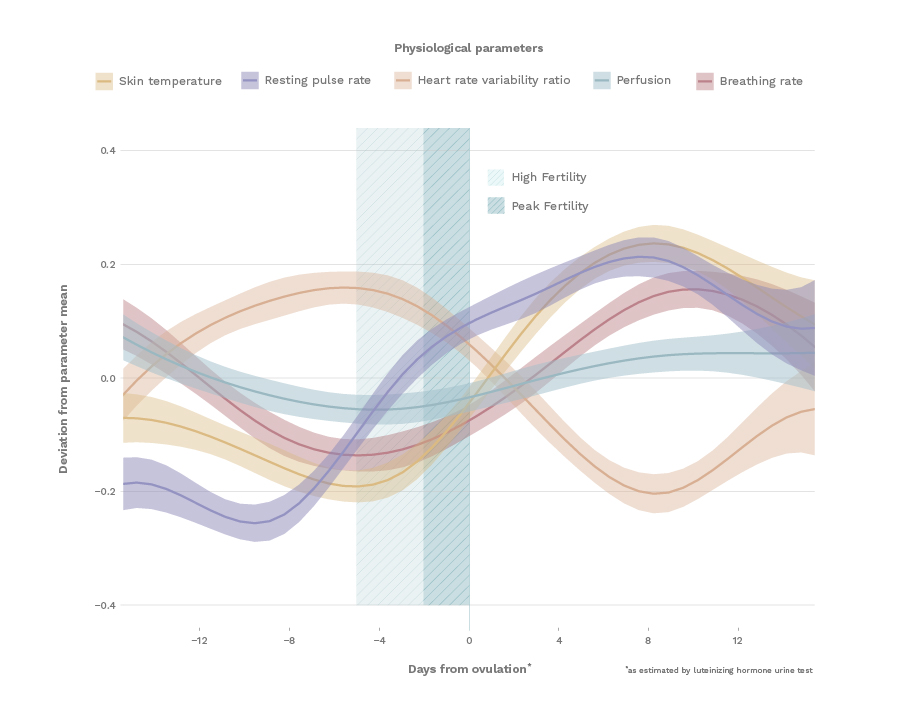
Ava Fertility has been shown in clinical studies to detect the fertile window in real time. Results are published in the peer-reviewed Journal of Medical Internet Research and confirm changes to five physiological signals of fertility throughout the menstrual cycle. Ava Fertility tracks all five.
.
Ava’s clinical science team is actively engaged in research related to women’s healthcare across their entire reproductive lifetime, including the topics of:

Revealing actionable, accurate data unique to every user.
Ava’s sensor bracelet collects data 25 times per second while the user sleeps. This information is then analyzed to pinpoint her fertile window in real time using multiple algorithms.
Our team of data scientists develop the advanced algorithms that drive Ava’s technology, processing the data collected from our users and applying machine-learning algorithms to provide the most accurate results for the broadest range of women’s cycles.
Fully anonymized user information.
Ava abides by the General Data Protection Regulation (GDPR), the highest security standard in both the European Union and the United States, and employs region-specific servers for data storage through Amazon Web Services (AWS). All user data is stored anonymously with the individual’s user ID, registration info, and other personally identifiable information (PII) stored separately. Find more information on the stringent security measures required by AWS and GDPR:

Takes the skin temperature at the wrist
Measures movement and determines sleep stage
Captures heart rate variability, pulse rate, breathing rate, and skin perfusion
Ava works with industry leaders in wearable technology, women’s health, and data science.
Deputy Head, Head Clinical Research, Department of Reproductive Endocrinology University of Zurich, Switzerland
Chief of Clinical Genetics, Feinberg School of Medicine, Northwestern University, Chicago, USA
Physician, Obstetrics & Gynecology, Kaiser Permanente Medical Center, Santa Clara, USA
Consultant in Community Gynaecology and Reproductive Healthcare, New Croft Centre, Newcastle, UK
Professor Emeritus, Department of Gynaecology and Obstetrics, University of Basel, Switzerland
Professor and Director, Division of Reproductive Endocrinology and Infertility University of Utah, Salt Lake City, USA
Chief, Division of Reproductive Endocrinology & Infertility, Columbia University, New York City, USA
